- Tel: 858.663.9055
 Email: info@nsjbio.com
Email: info@nsjbio.com
- Tel: 858.663.9055
- Email: info@nsjbio.com
Phospho antibody products are used to detect proteins modified by phosphorylation, one of the most important post-translational modifications in cell signaling. Phosphorylation can regulate enzyme activity, receptor activation, protein localization, protein stability, and interactions between signaling molecules. Because phosphorylation is often dynamic and stimulus-dependent, phospho-specific antibodies are valuable tools for studying pathway activation in cancer biology, stress response, growth factor signaling, immune signaling, and drug treatment models.
NSJ Bioreagents offers a broad selection of phospho antibody products for detecting phosphorylated targets in research applications such as western blot, immunohistochemistry, immunofluorescence, flow cytometry, and related assays.
Commonly studied phospho antibody targets include markers of growth factor signaling, cellular stress response, translation regulation, and DNA damage. These phosphorylation-specific antibodies are widely used to monitor pathway activation and evaluate experimental treatments.
Key targets include:
These targets provide a comprehensive view of signaling pathways controlling cell growth, survival, stress response, and genome stability.
A phosphorylation antibody is designed to recognize a protein only when a specific amino acid residue is phosphorylated, such as phospho-serine, phospho-threonine, or phospho-tyrosine sites. This makes phospho antibody detection different from total protein detection. A total protein antibody measures expression level, while a phospho-specific antibody helps determine whether a signaling pathway is active.
Examples include phospho-EGFR antibodies for receptor tyrosine kinase activation, phospho-EIF2 alpha antibodies for cellular stress signaling, and phospho-EIF4E antibodies for growth factor-induced translation activation. These reagents help researchers compare treated and untreated samples, evaluate inhibitor effects, and monitor stimulus-dependent signaling changes.
Phosphorylation-specific antibodies are widely used in cancer research because many oncogenic pathways are controlled by kinase signaling. Receptor tyrosine kinases, MAPK pathway proteins, PI3K-AKT pathway components, transcription factors, and translation regulators can all be studied through site-specific phosphorylation events.
Phospho antibody detection is also important for cellular stress response research. Targets such as phospho-EIF2 alpha (Ser51) provide insight into integrated stress response activation and translational repression, while phospho-EIF4E (Ser209) reflects growth signaling and cap-dependent translation regulation. Together, these markers help define how cells shift protein synthesis in response to stress, nutrients, growth factors, and drug exposure.
When selecting a phospho antibody, it is important to consider the exact phosphorylation site, target protein, species, application, and sample treatment conditions. Many phospho signals are transient, so sample handling, stimulation timing, lysis buffer composition, and phosphatase inhibition can strongly affect results. For western blot studies, paired untreated and stimulated samples can help demonstrate phosphorylation-dependent detection. For tissue or cell imaging, phospho antibodies can help localize active signaling within specific cellular compartments or tumor regions.
Researchers may also pair a phospho-specific antibody with a total protein antibody to distinguish changes in phosphorylation from changes in total protein abundance.
A broad selection of phospho antibody products is shown below, including phosphorylation-specific antibodies for signaling, cancer, stress response, kinase pathway, and translational regulation research.
These antibodies are part of a broader antibody panel offered by NSJ Bioreagents.
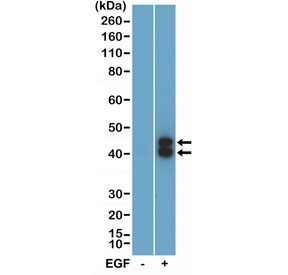
Phospho-ERK1/2 Antibody WB. Western blot analysis of human A431 cell lysates untreated (-) or stimulated (+) with epidermal growth factor (EGF) using a phospho-ERK1/2 antibody detecting ERK1 and ERK2 phosphorylated at Thr202 and Tyr204. Two bands are observed at approximately 42 kDa and 43 kDa, corresponding to ERK1 and ERK2, with increased signal following EGF treatment, demonstrating activation of MAPK signaling.
|
| ||

|
|
| ||

|